If you have diabetes and are active on social media lately, you've probably seen people claiming Ozempic, Wegovy or Zepbound as miracle drugs for their transformation. Not surprisingly, you might have also asked yourself the same question: could any of this actually help me?
The honest answer is: it depends. GLP-1 (glucagon-like peptide-1) drugs, such as semaglutide (Ozempic, Wegovy, Rybelsus) and tirzepatide (Mounjaro, Zepbound), have shown some weight loss and cardiometabolic benefits in clinical trials. But to make informed decisions, you need to know which uses are approved, what early research shows and where the risks lie.
What Are GLP-1 and GIP Medications, Exactly?
Glucagon-like peptide-1 (GLP-1) and Glucose-dependent insulinotropic polypeptide (GIP) are metabolic hormones released by our gut after we eat a meal. Both hormones help stimulate insulin secretion, but GLP-1 also inhibits glucagon release during hyperglycemia, slows gastric emptying and promotes satiety. In contrast, GIP has little effect on gastric emptying and a much weaker effect on suppressing appetite than GLP-1 [1. 2].
GLP-1 and dual GIP/GLP-1 receptor agonists are drug classes that mimic the function of the respective hormones. Semaglutide (Ozempic, Wegovy) is a GLP-1 receptor agonist, while Tirzepatide (Mounjaro, Zepbound) binds with both receptors (GLP-1 and GIP) and thus tends to have more effects on weight loss than semaglutide alone. Older members of the class include liraglutide, dulaglutide and exenatide [1, 3].
FDA Approval of GLP-1 and dual GIP/GLP-1 Drugs

If You Have Type 2 Diabetes
GLP-1 and dual GIP/GLP-1 medications are FDA-approved adjuncts for type 2 diabetic patients. They can be used alongside diet and exercise. The 2026 Standards of Care by the American Diabetes Association (ADA) recommend them for adults with type 2 diabetes who also have obesity or overweight, atherosclerotic cardiovascular disease, heart failure, chronic kidney disease or metabolic-associated steatotic liver disease [4].
Semaglutide also has an FDA label indication for reducing major adverse cardiovascular events in type 2 diabetic patients with cardiovascular disease. That is not a secondary benefit; it is on the label [5].
Clinical Evidence
In a 2021 clinical trial (SURPASS-2), tirzepatide was found to have greater reductions in hemoglobin A1c (HbA1c) and body weight than semaglutide 1 mg in people with type 2 diabetes [6]. Similar findings were reported in a 2025 real-world analysis of over ten thousand patients: tirzepatide lowered HbA1c by 1.3% and body weight by 10.2 kg, compared with 0.9% and 6.1 kg with semaglutide after 12 months [7].
A head-to-head clinical trial (SURMOUNT-5) published in The New England Journal of Medicine in 2025 also demonstrated the superiority of tirzepatide over semaglutide. Participants receiving tirzepatide achieved greater weight-loss milestones than those receiving semaglutide [3]. A 2025 meta-analysis has also reported a similar trend after analyzing clinical trials and observational studies: tirzepatide produced about 4% greater weight loss than semaglutide [8].
If You Have Type 1 Diabetes
Up to now, the FDA has not approved any GLP-1 or dual GIP/GLP-1 drugs for treating type 1 diabetes. The label on these medications, for example, Wegovy, clearly states that the drug has not been evaluated in patients with type 1 diabetes or in combination with insulin [9]. The label has not changed yet, but what has changed are the clinical recommendations around them.
For the first time, the 2026 ADA Standards of Care guidelines suggests that physicians may consider GLP-1 medications in selected patients with type 1 diabetes and obesity. However, the recommendation also stresses close monitoring, gradual reductions in insulin doses, maintaining adequate carbohydrate intake, ketone testing, sick-day rules and careful dose titration [4].
Clinical Evidence
We currently have very limited evidence in support of the use of GLP-1 or dual GIP/GLP-1 medications in treating adults with type 1 diabetes. Recently, a 26week trial (ADJUST-T1D) randomized 72 type 1 diabetic patients with obesity to semaglutide or placebo. Both groups used automated insulin delivery (AID). After the trial period, 36% of participants on semaglutide met a composite goal of ≥70% timeinrange, minimal hypoglycemia and ≥5% weight loss, versus 0% on placebo. The authors also reported modest HbA1c reductions (~0.3%) and an average 9 kg weight loss. No diabetic ketoacidosis (DKA) events occurred [10].
A separate double-blind crossover trial published in Nature Medicine found that weekly semaglutide increased time-in-range by 4.8% without increasing hypoglycemia. Although there was no incidence of a DKA event, two participants experienced recurrent euglycemic ketosis without acidosis [11]. Real-world retrospective analysis of data from the Barbara Davis Center compared type 1 diabetes patients on semaglutide and tirzepatide off-label over one year: 9.1% vs 21.4% body weight reduction and HbA1c reductions of 0.54% vs 0.68%, respectively [12].
However, major limitations of these trials are that they used advanced AID technology, included a small number of participants and had a short follow-up. So, even though emerging evidence looks promising, we still need to go a long way to establish these drugs as safe and effective treatment options for type 1 diabetic patients.
Safety Information and Red Flags
GLP-1 receptor agonists and dual GIP/GLP-1 receptor agonists do carry some risks even when used appropriately. Although most side effects are manageable, there are some rare but serious risks.
Acute pancreatitis
In January 2026, the UK MHRA has instructed manufacturers to update the product information for all GLP-1 and dual GIP/GLP-1 receptor agonists to highlight the potential risk of severe acute pancreatitis associated with these drugs, including rare reports of necrotizing and fatal pancreatitis [13].
Diabetic ketoacidosis
In patients with insulindependent diabetes, the risk of DKA is the primary concern. The UK MHRA has issued a specific safety warning that GLP-1 and dual GIP/GLP-1 medications are not substitutes for insulin. Insulin should never be stopped abruptly and should only be reduced stepwise with close monitoring. Since these products cause nausea or vomiting, they can lead patients or clinicians to reduce or stop insulin due to missed meals. Ultimately, this creates conditions for ketone accumulation even when glucose looks relatively normal [14].
Other side effects
The risk of hypoglycemia is generally low when these drugs are used alone or with metformin but rises when combined with insulin or sulfonylureas. Other side effects include gastrointestinal adverse effects, dehydration-related acute kidney injury, worsening diabetic retinopathy in some patients, aspiration risk during general anesthesia and gallbladder disease [5, 9, 15].
- Persistent Nausea, vomiting or diarrhea can cause fluid loss and acute kidney injury in susceptible patients.
- First observed in the SUSTAIN-6 trial, Rapid HbA1c reduction has been associated with worsening of pre-existing diabetic retinopathy [16]. Semaglutide labels warn about diabetic retinopathy complications, particularly in patients with pre-existing retinopathy [5, 9].
- Rapid weight loss raises cholelithiasis risk. Both semaglutide and tirzepatide labels flag this [9, 15].
- Newer labels such as Tirzepatide also warn about pulmonary aspiration risk during anesthesia because these drugs can delay stomach emptying [15].
Absolute Contraindications
Do not use GLP-1 and dual GIP/GLP-1 medications if you have any of the following:
- A personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN2)
- A history of pancreatitis or symptomatic gastroparesis
- Severe gastrointestinal disease
- Pregnant or planning pregnancy
Final Thoughts
GLP1 and dual GIP/GLP1 receptor are now widely used to help manage type 2 diabetes and obesity. They can lower HbA1c and support weight loss, and some agents also have proven cardiovascular benefits. For Type 1 diabetes, the evidence is still emerging. The 2026 ADA guidelines suggest a promising role for these drugs in selected adults with type 1 diabetes and obesity. For now, they may be used as off-label adjuncts, not as insulin replacements.
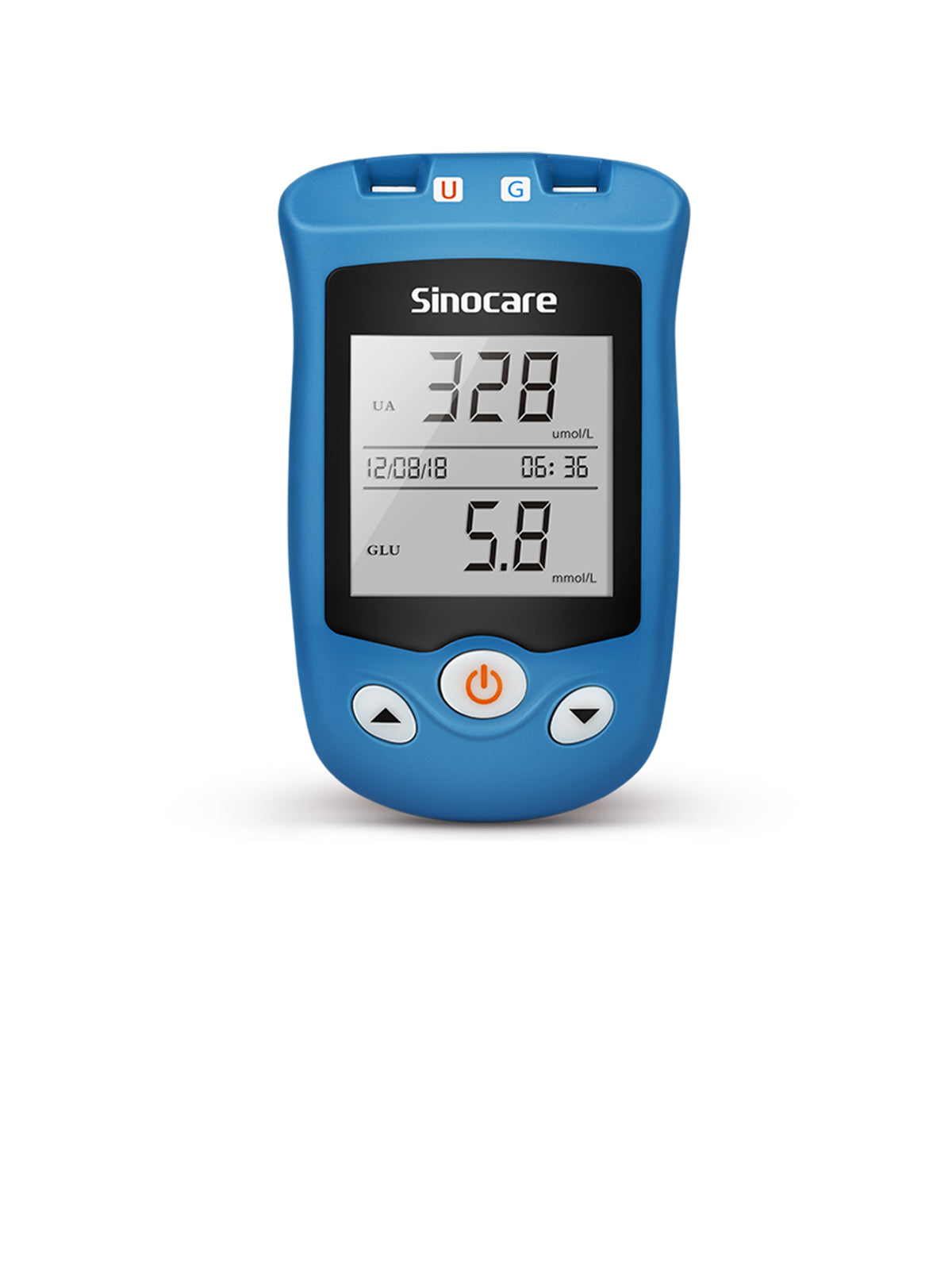
If you are starting semaglutide or tirzepatide, what’s important is tracking your readings consistently, as your blood sugar levels may spike or dip while doses and other diabetes medications are being adjusted. The Sinocare Safe AQ Smart blood glucose meter delivers results in 5 seconds using only 0.6 μL blood sample, making it easy to stay on top of those changes.
About Sinocare
Sinocare Inc., founded in 2002 in Changsha, China, is one of the world’s largest blood glucose monitor manufacturers. Our devices are used by more than 25 million people across 187 countries, and over 50% of Chinese people who selfmonitor their blood glucose rely on Sinocare products.
Sinocare’s Safe AQ Pro I meter includes a blood ketone testing reminder when glucose readings are high, which may prompt timely ketone checking and help avoid DKA.
Disclaimer: Educational content only. This article is not medical advice. Always consult your healthcare provider before starting, stopping or changing any medication.
References
1. Zheng Z, Zong Y, Ma Y, Tian Y, Pang Y, Zhang C, Gao J. Glucagon-like peptide-1 receptor: mechanisms and advances in therapy. Signal transduction and targeted therapy. 2024 Sep 18;9(1):234.
2. Yamanouchi D. The Roles of Incretin Hormones GIP and GLP-1 in Metabolic and Cardiovascular Health: A Comprehensive Review. International journal of molecular sciences. 2025 Dec 19;27(1):27.
3. Aronne LJ, Horn DB, le Roux CW, Ho W, Falcon BL, Gomez Valderas E, Das S, Lee CJ, Glass LC, Senyucel C, Dunn JP. Tirzepatide as compared with semaglutide for the treatment of obesity. N Engl J Med. 2025 Jul 3;393(1):26-36.
4. American Diabetes Association. Standards of care in diabetes—2026 [Internet]. Diabetes Care. 2026;49(Suppl 1):S1-S371. Available from: https://sentucuman.com.ar/docs/standards-of-care-2026.pdf.
5. Novo Nordisk A/S. Ozempic (semaglutide) injection, for subcutaneous use: prescribing information [Internet]. Bagsvaerd (Denmark): Novo Nordisk A/S; 2025 Oct Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/209637s035%2C209637s037lbl.pdf.
6. Frías JP, Davies MJ, Rosenstock J, Pérez Manghi FC, Fernández Landó L, Bergman BK, Liu B, Cui X, Brown K. Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes. New England Journal of Medicine. 2021 Aug 5;385(6):503-15.
7. Hoog MM, Vallarino C, Maldonado JM, Grabner M, Teng CC, Terrell K, Richard EL. Real-World effectiveness of Tirzepatide versus semaglutide on HbA1c and weight in patients with type 2 diabetes. Diabetes Therapy. 2025 Nov;16(11):2237-56.
8. Aamir AB, Latif R, Alqoofi JF, Almarzoq FA, Fallatah JO, Hassan GA, Saab FA. Comparative efficacy of tirzepatide vs. semaglutide in reducing body weight in humans: a systematic review and meta-analysis of clinical trials and real-world data. Journal of Clinical Medicine Research. 2025 May 13;17(5):285.
9. Novo Nordisk A/S. Wegovy (semaglutide) injection, for subcutaneous use: prescribing information [Internet]. Silver Spring (MD): U.S. Food and Drug Administration; 2025. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/215256s024lbl.pdf.
10. Shah VN, Akturk HK, Kruger D, Ahmann A, Bhargava A, Bakoyannis G, Pyle L, Snell-Bergeon JK. Semaglutide in adults with type 1 diabetes and obesity. NEJM evidence. 2025 Jul 22;4(8):EVIDoa2500173.
11. Pasqua MR, Tsoukas MA, Kobayati A, Aboznadah W, Jafar A, Haidar A. Subcutaneous weekly semaglutide with automated insulin delivery in type 1 diabetes: a double-blind, randomized, crossover trial. Nature medicine. 2025 Apr;31(4):1239-45.
12. Snell-Bergeon JK, Kaur G, Renner D, Akturk HK, Beatson C, Garg SK. Effectiveness of semaglutide and tirzepatide in overweight and obese adults with type 1 diabetes. Diabetes Technology & Therapeutics. 2025 Jan;27(1):1-9.
13. Medicines and Healthcare products Regulatory Agency (MHRA). GLP-1 receptor agonists and dual GLP-1/GIP receptor agonists: strengthened warnings on acute pancreatitis, including necrotising and fatal cases [Internet]. London: MHRA; 2026. Available from: https://www.gov.uk/drug-safety-update/glp-1-receptor-agonists-and-dual-glp-1-slash-gip-receptor-agonists-strengthened-warnings-on-acute-pancreatitis-including-necrotising-and-fatal-cases.
14. Medicines and Healthcare products Regulatory Agency (MHRA). GLP-1 receptor agonists: reports of diabetic ketoacidosis when concomitant insulin was rapidly reduced or discontinued [Internet]. London: MHRA; 2019. Available from: https://www.gov.uk/drug-safety-update/glp-1-receptor-agonists-reports-of-diabetic-ketoacidosis-when-concomitant-insulin-was-rapidly-reduced-or-discontinued.
15. Eli Lilly and Company. Mounjaro (tirzepatide) injection, for subcutaneous use: prescribing information [Internet]. Silver Spring (MD): U.S. Food and Drug Administration; 2025. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/215866s031lbl.pdf.
16.Husain M, Birkenfeld AL, Donsmark M, Dungan K, Eliaschewitz FG, Franco DR, Jeppesen OK, Lingvay I, Mosenzon O, Pedersen SD, Tack CJ. Oral semaglutide and cardiovascular outcomes in patients with type 2 diabetes. New England Journal of Medicine. 2019 Aug 29;381(9):841-51.









Leave a comment
All comments are moderated before being published.
This site is protected by hCaptcha and the hCaptcha Privacy Policy and Terms of Service apply.